From carbonyl compounds
Enroll to start learning
You’ve not yet enrolled in this course. Please enroll for free to listen to audio lessons, classroom podcasts and take practice test.
Interactive Audio Lesson
Listen to a student-teacher conversation explaining the topic in a relatable way.
Catalytic Hydrogenation
🔒 Unlock Audio Lesson
Sign up and enroll to listen to this audio lesson

Today we'll discuss how we can convert carbonyl compounds into alcohols. One of the primary methods is known as catalytic hydrogenation. Can anyone tell me what this process involves?

Isn't it when we add hydrogen to the carbonyl compound in the presence of a catalyst like platinum or nickel?

Exactly! In catalytic hydrogenation, we use a metal catalyst to enable the addition of hydrogen atoms to the carbonyl group. This process is crucial in organic synthesis. What products do you expect from this reaction?

If we use an aldehyde, we'll get a primary alcohol, right?

And if it's a ketone, we will get a secondary alcohol!

Correct! So, the type of alcohol formed depends on the carbonyl compound used in the reaction. Remember, aldehydes lead to primary alcohols and ketones produce secondary alcohols.
Reducing Agents: NaBH4 and LiAlH4
🔒 Unlock Audio Lesson
Sign up and enroll to listen to this audio lesson

Another effective way to reduce carbonyls is by using reducing agents. Can anyone name a couple of them?

Sodium borohydride and lithium aluminium hydride are two methods.

That's right! Sodium borohydride is often used to reduce aldehydes and ketones selectively to alcohols under milder conditions. On the other hand, lithium aluminium hydride is a stronger reducing agent—can anyone tell me what it can do to carboxylic acids?

LiAlH4 can reduce carboxylic acids to primary alcohols!

Exactly. However, we must handle LiAlH4 carefully as it reacts violently with water. Remember to always add it in dry solvents. What about the products we get from NaBH4?

We get primary and secondary alcohols based on whether we start from aldehydes or ketones.

Spot on! Understanding these methods and their effectiveness is key in organic synthesis. Do you see how this knowledge can be applied in real-life scenarios?
Importance and Applications
🔒 Unlock Audio Lesson
Sign up and enroll to listen to this audio lesson

Now that we understand how to prepare alcohols from carbonyl compounds, let’s discuss their importance. Why do you think alcohols are significant in both chemistry and industry?

They are used as solvents and in making many organic compounds!

Also, they play a vital role in biochemical processes too!

Absolutely! Alcohols are indeed crucial. Let’s summarize our key points from today: we can reduce carbonyls using hydrogen in catalytic hydrogenation or using specific reducing agents like NaBH4 and LiAlH4. Can anyone recall the types of alcohols produced from aldehydes and ketones again?

Aldehydes yield primary alcohols while ketones yield secondary alcohols!

Excellent! That's the gist of today's lesson.
Introduction & Overview
Read summaries of the section's main ideas at different levels of detail.
Quick Overview
Standard
The section discusses methods for preparing alcohols from carbonyl compounds, focusing on catalytic hydrogenation and the use of reducing agents like sodium borohydride and lithium aluminium hydride. It illustrates the transformations of aldehydes and ketones into primary and secondary alcohols respectively.
Detailed
In this section, we explore how carbonyl compounds, specifically aldehydes and ketones, can be converted into alcohols through reduction. The two major methods of reduction discussed are catalytic hydrogenation, where hydrogen is added in the presence of a metal catalyst like platinum or nickel, and the use of reducing agents such as sodium borohydride (NaBH4) and lithium aluminium hydride (LiAlH4). This process is significant in organic chemistry, as it highlights the transformation of functional groups, leading to the formation of alcohols, which can further react or serve as precursors for various biochemical processes and industrial applications. For example, aldehydes yield primary alcohols while ketones yield secondary alcohols upon reduction.
Youtube Videos









Audio Book
Dive deep into the subject with an immersive audiobook experience.
Reduction of Aldehydes and Ketones
Chapter 1 of 3
🔒 Unlock Audio Chapter
Sign up and enroll to access the full audio experience
Chapter Content
Alcohols are prepared by the reduction of aldehydes and ketones by addition of hydrogen in the presence of catalysts (catalytic hydrogenation). The usual catalyst is a finely divided metal such as platinum, palladium or nickel. It is also prepared by treating aldehydes and ketones with sodium borohydride (NaBH4) or lithium aluminium hydride (LiAlH4). Aldehydes yield primary alcohols whereas ketones give secondary alcohols.
Detailed Explanation
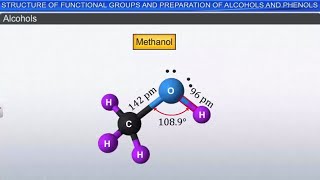
In this section, we learn how alcohols can be synthesized by reducing aldehydes or ketones. When an aldehyde undergoes reduction, it gains hydrogen atoms, resulting in a primary alcohol. For example, if we take formaldehyde (an aldehyde) and add hydrogen, we produce methanol (a primary alcohol). On the other hand, when a ketone is reduced, it forms a secondary alcohol. This is due to the difference in the molecular structure of aldehydes and ketones, where aldehydes have one carbon atom bonded to the functional group, while ketones have two. The resulting alcohols depend on the structure of the original carbonyl compound.
Examples & Analogies
Think of reducing an aldehyde like filling up a balloon. An aldehyde has one end open (the carbonyl group) and by adding hydrogen, you’re closing that end up to form a stable structure (an alcohol). For example, turning a half-filled cup (an aldehyde) into a full cup (the resulting alcohol) through the addition of more liquid (hydrogen).
Reduction of Carboxylic Acids and Esters
Chapter 2 of 3
🔒 Unlock Audio Chapter
Sign up and enroll to access the full audio experience
Chapter Content
Carboxylic acids are reduced to primary alcohols in excellent yields by lithium aluminium hydride, a strong reducing agent. However, LiAlH4 is an expensive reagent, and therefore, used for preparing special chemicals only. Commercially, acids are reduced to alcohols by converting them to the esters, followed by their reduction using hydrogen in the presence of a catalyst (catalytic hydrogenation).
Detailed Explanation
This part discusses how carboxylic acids, which are stronger acids than aldehydes or ketones, can also be transformed into alcohols. The process involves a strong reducing agent, lithium aluminium hydride (LiAlH4). This powerful reagent does a fantastic job of attaching more hydrogen to carboxylic acids, effectively converting them into primary alcohols. However, since LiAlH4 is quite costly, in industrial settings, it's often more economical to first convert the acid to an ester and then reduce that ester using hydrogen, which serves the same purpose at a lower cost.
Examples & Analogies
Consider a car being heavily weighed down, representing a carboxylic acid. To lighten the weight (reduce) and enhance its performance (convert to alcohol), you might first unload it (convert to an ester) before giving it a speed boost (adding hydrogen) so that it can function better on the road.
Synthesis Using Grignard Reagents
Chapter 3 of 3
🔒 Unlock Audio Chapter
Sign up and enroll to access the full audio experience
Chapter Content
Alcohols are produced by the reaction of Grignard reagents with aldehydes and ketones. The first step of the reaction is the nucleophilic addition of Grignard reagent to the carbonyl group to form an adduct. Hydrolysis of the adduct yields an alcohol.
Detailed Explanation
Grignard reagents are a special class of compounds that serve as strong nucleophiles. Here, they play a crucial role in synthesizing alcohols by attacking the carbonyl carbon in aldehydes or ketones. This reaction creates a new intermediate compound (the adduct) that can then undergo hydrolysis, ultimately yielding an alcohol. This method is particularly useful in organic synthesis due to its versatility and effectiveness.
Examples & Analogies
Imagine you’re building a LEGO tower (the aldehyde or ketone). When you bring in a special piece (the Grignard reagent), it locks into your structure (attacks the carbonyl), allowing you to add even more layers (hydrolysis) on top, resulting in a more complex and tall tower (the alcohol).
Key Concepts
-
Catalytic Hydrogenation: A method to add hydrogen across the carbonyl to convert it into alcohol.
-
Sodium Borohydride: A reducing agent useful in converting carbonyls to alcohols.
-
Lithium Aluminium Hydride: A powerful reducing agent that can reduce a wider range of carbonyl compounds.
Examples & Applications
Example of catalytic hydrogenation with butanal to produce butanol.
Using sodium borohydride to reduce acetone to isopropanol.
Memory Aids
Interactive tools to help you remember key concepts
Rhymes
When carbonyls reduce, alcohols seduce, adding H2 will produce!
Stories
Imagine a bustling factory where hydrogen atoms come in with help, transforming carbonyl compounds into the life of the party - alcohols.
Memory Tools
AH - Aldehydes produce Hydros (Primary Alcohols); K - Ketones yield Secondary - Just remember AH-K!
Acronyms
HARP
Hydrogen
Aldehydes
Reduce
Produce (Alcohols).
Flash Cards
Glossary
- Catalytic Hydrogenation
A process where hydrogen is added to a compound in the presence of a catalyst to reduce a carbonyl group to an alcohol.
- Sodium Borohydride
A mild reducing agent used to convert aldehydes and ketones into alcohols.
- Lithium Aluminium Hydride
A strong reducing agent capable of reducing carboxylic acids to primary alcohols, as well as aldehydes and ketones.
Reference links
Supplementary resources to enhance your learning experience.
