Reactions involving cleavage of carbon – oxygen (C–O) bond in alcohols
Enroll to start learning
You’ve not yet enrolled in this course. Please enroll for free to listen to audio lessons, classroom podcasts and take practice test.
Interactive Audio Lesson
Listen to a student-teacher conversation explaining the topic in a relatable way.
C–O Bond Cleavage in Alcohols
🔒 Unlock Audio Lesson
Sign up and enroll to listen to this audio lesson

Today, we will discuss the reactions involving the cleavage of carbon-oxygen bonds in alcohols. Can someone explain what happens during the reaction of alcohols with hydrogen halides?

When alcohols react with hydrogen halides, they form alkyl halides and water, right?

Exactly! This reaction is a nucleophilic substitution where the proton from the hydrogen halide leads to the formation of a carbocation. What do you think influences the rate of this reaction?

I believe the class of alcohol is important—tertiary alcohols react faster because they form more stable carbocations, don't they?

Correct! Tertiary alcohols form more stable carbocations, while primary alcohols react more slowly. Remember the Lucas test which helps distinguish between them. Can anyone summarize?

The reactivity of alcohols with hydrogen halides depends on whether they are primary, secondary, or tertiary.

Great summary! Alcohols can also undergo further reactions, such as with phosphorus trihalides.
Dehydration of Alcohols
🔒 Unlock Audio Lesson
Sign up and enroll to listen to this audio lesson

Next, let's discuss dehydration of alcohols. Who can tell me the product formed from dehydration?

Ethanol can be dehydrated to form ethene.

Correct! Dehydration reactions typically produce alkenes. Which alcohols do you think dehydrate the easiest?

Tertiary alcohols would dehydrate easier than primary because they form more stable carbocations!

Precisely! The dehydration order follows Tertiary > Secondary > Primary. Can anyone provide an example of a mild dehydrating condition?

Using concentrated sulfuric acid at a lower temperature could promote alkene formation.

Exactly! Dehydration is essential in producing alkenes in organic synthesis.
Acidity of Alcohols and Phenols
🔒 Unlock Audio Lesson
Sign up and enroll to listen to this audio lesson

Now let’s dive into the acidity of alcohols and phenols. How do we compare the acidity of these two classes?

I think phenols are generally more acidic than alcohols because the phenoxide ion is stabilized by resonance.

Great insight! The resonance delocalization in phenoxide ions contributes to their stability. What about substituents—how can they influence acidity?

Electron-withdrawing groups increase acidity by stabilizing the phenoxide ion, while electron-donating groups do the opposite.

Exactly! That’s why nitrophenols are more acidic than cresols. Can someone summarize our findings on acidity?

Phenols are generally stronger acids than alcohols and substituents can either increase or decrease acidity.
Cleavage of C–O Bonds in Ethers
🔒 Unlock Audio Lesson
Sign up and enroll to listen to this audio lesson

Let’s wrap up with ethers. Who remembers how ethers react in comparison to alcohols?

Ethers are less reactive, but they can be cleaved under extreme conditions using hydrogen halides.

Correct! Their C–O bonds are cleaved at high temperatures with excess hydrogen halides. Can anyone explain why ethers are less reactive?

Ethers lack the acidic O-H bond that alcohols and phenols have, so they don’t ionize as easily.

Good point! Understanding the reactivity differences helps in selecting compounds for synthesis.
Introduction & Overview
Read summaries of the section's main ideas at different levels of detail.
Quick Overview
Standard
In this section, we explore how the cleavage of C–O bonds occurs in alcohols primarily through reactions with hydrogen halides, phosphorus trihalides, and dehydration processes. It also discusses the acidic nature of alcohols and phenols, differences in reactivity among various classes of alcohols, and preparation methods for ethers.
Detailed
Reactions Involving Cleavage of C–O Bond
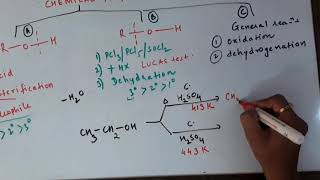
This section discusses the chemical reactions of alcohols, phenols, and ethers, particularly focusing on the cleavage of carbon-oxygen (C–O) bonds. Alcohols may react with hydrogen halides (HX) to produce alkyl halides through nucleophilic substitution. Reactivity varies according to the alcohol class: primary, secondary, or tertiary, which can be evaluated using the Lucas test.
Further, alcohols can also react with phosphorus trihalides to yield alkyl halides efficiently. Dehydration reactions convert alcohols into alkenes, with tertiary alcohols dehydrating more readily than secondary and primary alcohols.
Phenols exhibit acidic characteristics due to the resonance stabilization of their phenoxide ions when reacting with bases. The section highlights the increased acidity of phenols by substituents that withdraw electron density. Lastly, ethers, being less reactive, can cleave their C–O bonds under extreme conditions. All these reactions underpin the significance of C–O bond cleavages in organic synthesis and industry.
Youtube Videos


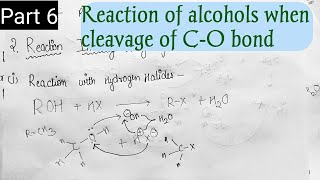








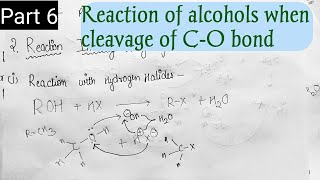







Audio Book
Dive deep into the subject with an immersive audiobook experience.
Cleavage of C–O Bond in Alcohols
Chapter 1 of 4
🔒 Unlock Audio Chapter
Sign up and enroll to access the full audio experience
Chapter Content
The reactions involving cleavage of C–O bond take place only in alcohols. Phenols show this type of reaction only with zinc.
Detailed Explanation
This point establishes a distinction between how alcohols and phenols behave in chemical reactions. Specifically, the cleavage of the carbon-oxygen (C–O) bond occurs in alcohols but is limited in phenols unless reacting with zinc. This sets the stage for understanding how alcohols can participate in a variety of reactions that phenols cannot.
Examples & Analogies
Think of alcohols as versatile tools in a toolbox — they can be used for many types of repairs (reacting in various ways), while phenols are like specialty tools that only work effectively for specific tasks (limited reactions).
Reactions with Hydrogen Halides
Chapter 2 of 4
🔒 Unlock Audio Chapter
Sign up and enroll to access the full audio experience
Chapter Content
- Reaction with hydrogen halides: Alcohols react with hydrogen halides to form alkyl halides. The difference in reactivity of three classes of alcohols with HCl distinguishes them from one another (Lucas test).
Detailed Explanation
When alcohols react with hydrogen halides (like HCl), they form alkyl halides — compounds where a halogen replaces the hydroxyl (–OH) group. This reaction showcases the reactivity differences among primary, secondary, and tertiary alcohols. For example, the Lucas test is a classic method to identify the alcohol class based on the speed at which turbidity appears when alcohol is mixed with Lucas reagent. Tertiary alcohols show immediate turbidity because they form halides faster than primary alcohols.
Examples & Analogies
Imagine making a smoothie with different fruits. Different fruits (alcohols) react differently with the blender (hydrogen halides). Tertiary fruits (tertiary alcohols) blend super fast, while primary fruits take longer to mix well.
Dehydration Reactions
Chapter 3 of 4
🔒 Unlock Audio Chapter
Sign up and enroll to access the full audio experience
Chapter Content
Dehydration: Alcohols undergo dehydration (removal of a molecule of water) to form alkenes on treating with a protic acid such as concentrated H2SO4 or H3PO4.
Detailed Explanation
In dehydration reactions, alcohols lose a water molecule through the influence of a strong acid. This process converts alcohols into alkenes, which are unsaturated hydrocarbons with double bonds. The ability to undergo dehydration strongly depends on the structure of the alcohol; tertiary alcohols dehydrate more easily than secondary or primary alcohols because of carbocation stability.
Examples & Analogies
It’s like squeezing juice out of a sponge. Here, the sponge (alcohol) releases water (dehydration) when pressure is applied (strong acid), and you end up with a concentrated juice (alkene).
Oxidation of Alcohols
Chapter 4 of 4
🔒 Unlock Audio Chapter
Sign up and enroll to access the full audio experience
Chapter Content
Oxidation: Oxidation of alcohols involves the formation of a carbon-oxygen double bond with cleavage of O-H and C-H bonds.
Detailed Explanation
Oxidation reactions for alcohols typically result in the formation of a carbon-oxygen double bond (like in aldehydes or ketones) and involve the breaking (cleavage) of the O-H and C-H bonds. The type of oxidation reaction (mild or strong) will dictate whether the products are aldehydes, ketones, or carboxylic acids, depending on whether a primary, secondary, or tertiary alcohol is being oxidized.
Examples & Analogies
Think of a fruit ripening. The process is akin to oxidation: here, ethylene gas (oxidizing agent) converts the fruit's natural compounds (alcohols) into different or more complex molecules (aromatic aldehydes, ketones, or acids) as it ages.
Key Concepts
-
Nucleophilic Substitution: A reaction where a nucleophile replaces a group in a molecule.
-
Carbocation Stability: Tertiary carbocations are more stable than secondary and primary.
-
Dehydration Reactions: Convert alcohols to alkenes by removing water.
-
Acid-Base Nature: Alcohols and phenols are acids, with varying strength affected by substituents.
Examples & Applications
Dehydration of ethanol produces ethene when heated.
The reaction of phenol with sodium hydroxide forms the phenoxide ion.
Memory Aids
Interactive tools to help you remember key concepts
Rhymes
In the land of alcoholics, we find, HBr converts with a twist to unwind.
Stories
Once upon a time, in a laboratory far away, alcohols reacted with halides to form halides every day!
Memory Tools
FAB: Forms Alkyl Bromides (for alcohol reactions with HBr).
Acronyms
A.B.C.
Acids
Bases
Cleavage - Key points of alcohol reactivity.
Flash Cards
Glossary
- Nucleophilic Substitution
A type of reaction where a nucleophile replaces a leaving group in a molecule.
- Carbocation
An organic molecule with a positively charged carbon atom.
- Dehydration
The removal of water from a molecule, often resulting in the formation of a double bond.
- Phenoxide Ion
The anion formed by deprotonation of phenol, stabilized by resonance.
- Acidic Strength
The tendency of a compound to donate protons, influenced by its molecular structure.
Reference links
Supplementary resources to enhance your learning experience.
